Bioprinting Breakthrough Offers New Hope for Low Back Pain Relief
Bioprinting Breakthrough Offers New Hope for Low Back Pain Relief
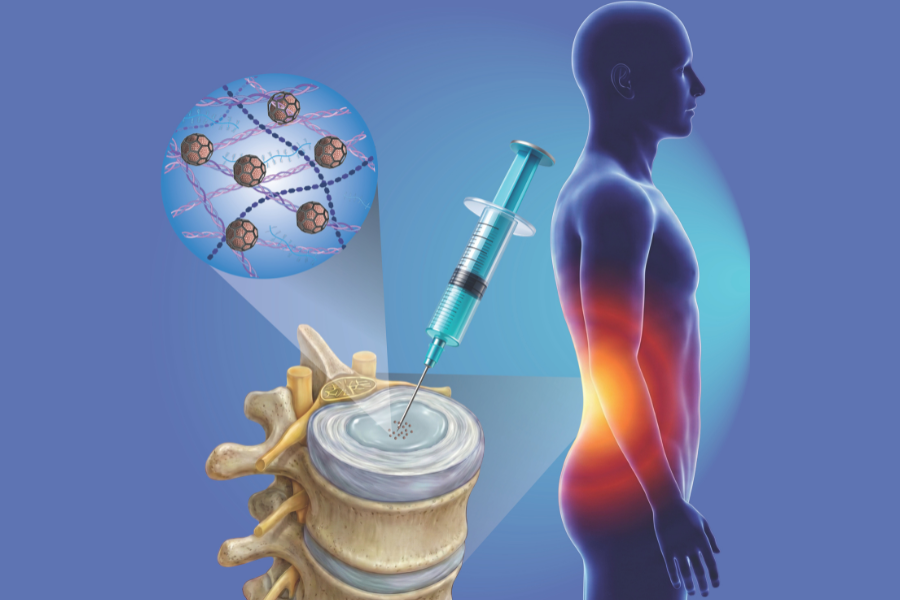
Innovative injectable cell therapy could one day repair worn-out spinal “shock absorbers” — powered by BIO INX’ DEGRAD INX, a cutting-edge biodegradable biomaterial. (Image credit to Rathina Vel Balasubramanian from TU Wien)
Low back pain affects around 80 % of people at some point in life, commonly traced back to damage in the intervertebral discs — the soft, gel-filled “shock absorbers” between the vertebrae in our spine. When these discs wear down or herniate, the cushioning motion is lost and painful pressure can be placed on nerves, severely limiting mobility and quality of life.
Current treatments typically focus on symptom relief, such as physiotherapy, or on altering the disc mechanically — for example shrinking the disc core to reduce pressure, which unfortunately also reduces its function. In severe cases, surgeons may resort to spinal fusion, locking vertebrae together to stop painful motion but permanently reducing flexibility and causing additional stress on the surrounding intervertebral discs .
Now, researchers from TU Wien — led by Prof. Aleksandr Ovsianikov — have published exciting new laboratory results pointing toward a fundamentally different approach: an injectable, cell-based therapy that could help regenerate disc tissue rather than simply manage pain.*
What Makes This New Approach So Promising?
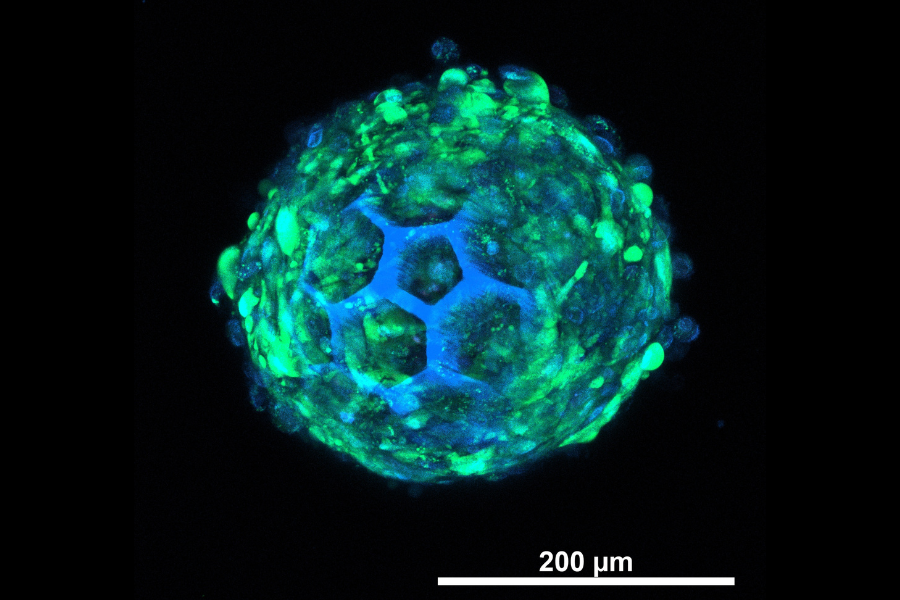
Instead of traditional surgery or purely symptom-focused treatment, the researchers used tiny cell clusters protected by a biodegradable cage — called scaffolded spheroids — designed for injection into damaged discs.
These scaffolded spheroids are built using high-resolution multiphoton lithography, a precision 3D printing method that lets scientists create miniature, geometrically complex structures at the micrometer scale. At the heart of this technology are biodegradable materials — including DEGRAD INX, BIO INX’s polyester-based resin — that act as biodegradable cages around the cell clusters. This approach has several important advantages:
🌱Protection during Injection: The cages protect cells from the shear forces experienced when injected through a needle, improving cell survival.
🚰Nutrient and Waste Transport: Their porous design allows nutrients and waste to pass freely, preventing core necrosis (cell death due to lack of nutrition).
🔄Tissue Formation: Cells within these cages can assemble into larger, mechanically robust tissues that start to resemble healthy disc cartilage.
💪Mechanical Strength: In lab studies, these constructs withstand mechanical loading that approaches the behavior of natural, healthy intervertebral discs — a key factor if this future therapy is to function in the dynamic environment of the spine.
💉Minimally Invasive: Because this method is injectable, it avoids the risks associated with major spinal surgery and could offer a much gentler treatment option in the future.
What Did the New Publication Find?
In the study published in ACS Applied Materials & Interfaces, researchers tested these scaffolded spheroids under conditions mimicking the disc environment. Their findings included:
✔️ High cell viability and extracellular matrix production — important for forming new functional tissue.
✔️ Cells differentiated into nucleus pulposus–like tissue, which is the inner core of a healthy disc.
✔️ The constructs retained structural integrity and cell function even after injection through a fine needle.
✔️ Mechanical properties of assembled tissue approached those of native human disc tissue, indicating the potential to sustain spinal loads.
These results suggest that this technology — combining advanced 3D micro-scaffolds with precise cell biology — could provide a realistic path toward regenerative treatments for degenerative disc disease. Further work, including preclinical tests, is still needed, but the data are compelling.
How DEGRAD INX Enables This Science
At the core of this breakthrough is DEGRAD INX, BIO INX’s biodegradable resin specially formulated for multiphoton lithography. It enables researchers to 3D-print precise, tiny, porous structures that:
- are biocompatible and degrade over time as new tissue forms,
- provide mechanical strength during the critical early phases of tissue development, and
- support nutrient flow and cellular self-organization essential for building functional tissue.
Without materials like DEGRAD INX — combining controlled biodegradability with high-resolution printability — such injectable, cell-protective scaffolds simply would not be possible.